Brain (1998), 121, 1155–1164

Differential effect of a dopaminergic agonist on prefrontal function in traumatic brain injury
patients

Sharon McDowell,1,2 John Whyte1,2 and Mark D’Esposito3
1Moss Rehabilitation Research Institute, 2Department of Correspondence to: Mark D’Esposito, Department of Physical Medicine and Rehabilitation, Temple University Neurology, University
of Pennsylvania Medical Center, School
of Medicine, 3Department of Neurology, University 3400 Spruce Street, Philadelphia, PA 19104-4283, USA. of Pennsylvania Medical Center, Philadelphia, PA, USA E-mail: despo@mail.med.upenn.edu
Summary
We examined the effects of low-dose bromocriptine,
a D2 dopamine receptor agonist,
on processes thought to be subserved by the prefrontal
cortex, including working memory and executive function,
in individuals with traumatic brain injury. A group of 24 subjects
was tested using a double-blind, placebo-controlled crossover
trial, counterbalanced for order. Bromocriptine
was
found
to improve performance on some tasks thought
to be subserved by prefrontal
function, but not others.
Specifically, there was improvement
in performance on
clinical measures of executive function
and in dual-task performance, but not measures that tap the ability to maintain information in working memory without
significant executive demands. Also, on control tasks not
thought to be dependent on the prefrontal
cortex, no improvement on bromocriptine
was noted. These results demonstrate a selective effect of bromocriptine
on cognitive processes which involve
executive control, and provide a foundation
for potential therapies for patients
with prefrontal damage causing dysexecutive syndromes.
Keywords: dopamine;
executive function; frontal lobes; traumatic
brain injury; working memory
Abbreviations: MANOVA = multivariate analysis
of variance; TBI = traumatic brain injury

Introduction
Executive function is a loosely defined term that is meant to
capture a wide range of cognitive
abilities such as planning,
set-shifting, sustaining and dividing attention,
organization and goal-integration. Numerous clinical neuropsychological measures, such as the Wisconsin
Card Sorting Test and the
Stroop Test (Lezak, 1995), have been designed
to tap this function, and patients
with lesions of the prefrontal cortex are often impaired on these
measures relative to patients with posterior lesions (e.g. Milner, 1963; Vendrell
et al., 1995). Also, some of the neuropsychological
measures which have been adapted for study during PET scanning have demonstrated activation of the prefrontal
cortex during performance by normal subjects (e.g. Bench et al.,
1993; Berman et al., 1995). Such empirical
evidence has provided a link between executive
processes and prefrontal
function.
More recently, investigators have sought to define
the cognitive processes that may be subserved by the prefrontal cortex more specifically. One such process is working memory, which refers
to the short-term storage of information
that is not accessible in the environment, and the set of processes that keep this information active
for later use in
© Oxford University Press 1998
behaviour. Baddeley (1992) has proposed a multicomponent
model of working memory. One component of this model, the ‘central executive
system’, analogous
to
Shallice’s
‘supervisory attentional system’ (Shallice, 1982), is thought to regulate the allocation of limited attentional
resources and to co-ordinate the manipulation of the information required in complex
or novel tasks.
Dual-task paradigms, which are thought
to tap these systems, have been demonstrated to be more impaired in patients with focal frontal
lesions (Baddeley et
al., 1997), as well
as frontal lesions secondary
to traumatic brain injury
(TBI) (Hartman et al.,
1992; Cicerone, 1996; McDowell
et al., 1997). Also,
a functional MRI
study
in normal subjects during
dual-task
performance
showed activation of the prefrontal cortex (D’Esposito et al., 1995). Recently, Kimberg and Farah (1993) have demonstrated that impairments on
a
wide
range
of
clinical
executive measures thought to tap prefrontal function
can be explained in a
computational
model
by
weakening
associations
in working memory without relying
on
a unique ‘central executive’. The neural correlates of working memory
have been extensively
studied
in
monkeys
and
humans
using
delayed response tasks. It has been found that lesions to the lateral prefrontal cortex impair
performance on these tasks
(Funahashi et al., 1993; Verin et al.,
1993), and the prefrontal
cortex is activated specifically
during the delay period of these
tasks (Fuster and Alexander, 1971; Funahashi et al.,
1989; Jonides et al., 1993; Zarahn et al., 1996).
Dopamine
seems
to
be
an
important neurotransmitter for prefrontal function. Non-human
primate studies have demonstrated that there are increased
concentrations of dopamine, dopamine receptors and dopamine-containing
terminals in the lateral prefrontal cortex (Brown et
al., 1979). Converging evidence suggests
that neurochemical alterations in the dopaminergic neurotransmitter system can cause frontal lobe dysfunction. Non-human primate studies have
demonstrated that dopamine depletion
(Sawaguchi and Goldman-Rakic,
1991) and pharmacological dopamine blockade (Brozoski et al., 1979), in addition
to focal damage to the striatum (Divac et al.,
1967), cause difficulty with spatial working memory tasks. Ageing, which decreases
D2 dopamine receptor levels
in
the
prefrontal cortex (Wong
et al., 1984; de Keyser et al.,
1990; Rinne et al., 1990) has also
been reported to impair spatial working memory in monkeys (Arnsten et al., 1995). In humans,
patients with Parkinson’s disease, who have dopamine
depletion from degeneration of the nigrostriatal pathway, exhibit
cognitive deficits attributable
to
prefrontal
dysfunction
(e.g.
Taylor et al., 1986).
Thus, the goal of this study was to investigate
further the link between dopamine and cognitive
processes thought to depend
on prefrontal function.
Based on the cognitive models summarized above, we investigated the effects of a dopaminergic agonist, bromocriptine, on (i) working
memory measures, (ii) clinical
measures of executive
function and (iii) measures
not thought to tap prefrontal function. We chose
patients with TBI, who we have previously demonstrated to have working memory
and executive impairments (McDowell et al., 1997).
These deficits are likely to be due to frontal lobe contusions (Adams et al., 1980) and diffuse axonal
injury that disrupts dopaminergic inputs to the prefrontal cortex and neural connections between the prefrontal cortex and
other areas of the brain (Adams
et al., 1982). Based on the work in non-human
primates, it was expected that performance on measures thought to depend on prefrontal
function would improve on bromocriptine.
Method
Subjects
We measured the cognitive
effects
of bromocriptine in
24
subjects (20 men, four women)
who had TBI causing concussion with loss of consciousness (Glasgow
Coma Scale
< 8) more than 4
weeks
prior
to
testing.
The
subjects’
median age was 32.5 years (range 15–55, with the exception of one 73-year-old patient) and mean duration
of education was 13
years
(range
9–18).
Subjects
were
excluded
for
dementia (Mini-Mental Status Exam score < 26), deficits in oculomotor, uncorrected acuity
or visual field, as noted on
medical records or by brief bedside examination, a major mental illness, uncontrolled high blood pressure, pregnancy,
or current use of centrally
acting medications. Based on neuroimaging studies that were available
to us, 18 out of 22
patients had evidence of frontal injury. We did not have any
radiology reports for two subjects, although one was noted
to have facial and orbital
fractures. Table 1 gives more clinical and demographic details
of our subjects. Twenty-one of the subjects in this study were also participants
in a previously reported study not involving
pharmacological treatment (McDowell et al.,
1997). The protocol
described here was approved
by the University of Pennsylvania IRB; all subjects gave informed consent prior to participation.
Experimental procedure
After a practice session
which was used to familiarize subjects with the tasks, and reduce the effects of practice during the actual test sessions, each subject was tested twice,
once on
2.5
mg bromocriptine and once on a placebo, in a randomized, double-blind crossover
design counterbalanced for the order
of drug administration. Testing was scheduled to
occur
90
min after oral pill administration, since the range of peak effectiveness for bromocriptine was estimated from previous
studies (Durso et al., 1982; Luciana et al.,
1992) to occur between 90 and 180 min after pill ingestion.
Cognitive tasks
All
of the tasks used in this study were thought
to depend on prefrontal
cortex function. Specifically, tasks were chosen
to probe different aspects of prefrontal cortex function, including (i) the process of maintenance
of information in working memory buffers
and (ii) executive
control processes, which were examined with an experimental measure as well as commonly
used clinical measures.
We also included the bi-letter cancellation task, a control task which was used
to test non-specific arousal, attention
and motor processes (Luciana et al., 1992; Lezak,
1995) and should not depend on
the prefrontal cortex. Except for the trailmaking test, a controlled oral word-association test (FAS Test) and the
control task, all tasks were performed on a portable
computer. The order of the tests was fixed in the order
in which they are
described below. Not all subjects completed all tests, because of time constraints
and the desire of one patient to discontinue the testing for social reasons; for these patients two lengthy and often frustrating
tests were omitted (the Verbal Span and Wisconsin
Card Sorting Test).
Dual-task paradigm
Subjects performed a simple visual reaction time task and then
performed it concurrently with each of two other tasks. In
the primary task, subjects responded
with a keypress to a
Table 1 Demographic and clinical information of the TBI patients
|
Age
(years)
|
Education
|
Sex
|
Race
|
Handedness
|
GCS
|
Aetiology
|
Lesions
|
Skull
|
|
26
|
13
|
M
|
W
|
R
|
6
|
MVA
|
R frontoparietal contusion
|
|
|
|
|
|
|
|
|
|
R basal ganglia contusion
|
|
|
|
|
|
|
|
|
|
R occipital/L brainstem contusion
|
|
|
43
|
15
|
M
|
B
|
R
|
UC
|
GSW to
L face
|
R frontal contusion
|
|
|
28
|
11
|
M
|
W
|
R
|
3
|
motorcycle
|
L frontoparietal/convexity SDH
|
|
|
|
|
|
|
|
|
accident
|
Diffuse R hemisphere oedema
|
+
|
|
55
|
12
|
M
|
W
|
R
|
UC
|
Hit by object
|
L frontoparietal SDH
|
|
|
|
|
|
|
|
|
falling 40'
|
Small L temporal SDH
|
+
|
|
73
|
12
|
F
|
W
|
R
|
|
Fell down steps
|
L frontoparietal SDH
|
|
|
53
|
13
|
M
|
W
|
|
|
MVA
|
R frontal contusion
|
|
|
|
|
|
|
|
|
|
R temporal contusion
|
|
|
31
|
13
|
F
|
W
|
R
|
DC
|
MVA
|
Bilateral frontal contusions
|
|
|
|
|
|
|
|
|
|
Diffuse oedema
|
|
|
28
|
12
|
M
|
W
|
L
|
UC
|
GSW to
R
|
R frontoparietal SDH
|
|
|
|
|
|
|
|
|
lateral orbit
|
Intraventricular oedema + mass effect
|
|
|
|
|
|
|
|
|
|
R occipital infarct/temporalcontusion
|
+
|
|
48
|
16
|
M
|
W
|
L
|
UC
|
MVA
|
Bilateral frontal contusion and SAH
|
+
|
|
28
|
16
|
M
|
W
|
R
|
UC
|
Aneursym
|
L ACoA
aneurysm/diffuse L oedema
|
|
|
54
|
13
|
M
|
|
|
UC
|
Assault?
|
L frontal SAH/subfrontal contusion
|
|
|
|
|
|
|
|
|
|
Diffuse oedema
|
|
|
43
|
12
|
M
|
W
|
R
|
CC
|
Pedestrian/
|
R inferior frontal shearing/frontal contusion
|
|
|
|
|
|
|
|
|
MVA
|
R basal ganglia/temporal SDH
|
|
|
|
|
|
|
|
|
|
Intraventricular SAH
|
|
|
|
|
|
|
|
|
|
L parietal contusion
|
|
|
39
|
15
|
M
|
W
|
R
|
5
|
Fell 20
ft
|
Bilateral frontal/basal ganglia decreased
|
|
|
|
|
|
|
|
|
|
perfusion, R occipital decreased perfusion
|
|
|
|
|
|
|
|
|
|
Bilateral temporal contusion
|
|
|
20
|
14
|
M
|
W
|
R
|
7
|
MVA
|
Corpus callosum shearing
|
|
|
|
|
|
|
|
|
|
R internal capsule decreased perfusion
|
|
|
|
|
|
|
|
|
|
L intraventricular haemorrhage
|
|
|
43
|
18
|
M
|
W
|
R
|
UC
|
MVA
|
Bilateral subfrontal hygromas/SAH
|
|
|
|
|
|
|
|
|
|
Diffuse SAH/intraventricular haemorrhage
|
|
|
24
|
15
|
M
|
H
|
R
|
3
|
MVA
|
Bilateral frontoparietal contusions
|
|
|
|
|
|
|
|
|
|
Corpus callosum haemorrhage
|
|
|
|
|
|
|
|
|
|
R basal ganglia/thalamus haemorrhage
|
|
|
|
|
|
|
|
|
|
Small L temporal haemorrhage
|
+
|
|
24
|
16
|
M
|
W
|
R
|
*
|
1000-ft fall
|
L parietal punctate haemorrhage
|
|
|
|
|
|
|
|
|
|
R temporal contusion
|
+
|
|
47
|
15
|
F
|
W
|
R
|
UC
|
MVA
|
R caudate head contusion
|
|
|
|
|
|
|
|
|
|
R parasaggital contusion
|
|
|
19
|
12
|
M
|
W
|
L
|
8
|
MVA
|
L frontal haemorrhage
|
|
|
|
|
|
|
|
|
|
R internal capsule, pons and
midbrain
|
|
|
|
|
|
|
|
|
|
haemorrhage, L medial temporal contusion
|
|
|
18
|
12
|
M
|
B
|
L
|
UC
|
MVA
|
L temporal SDH
|
+
|
|
34
|
17
|
M
|
W
|
R
|
3
|
Pedestrian/
|
R frontal SDH/contusions
|
|
|
|
|
|
|
|
|
MVA
|
|
|
|
15
|
9
|
F
|
W
|
L
|
UC
|
MVA
|
No X-rays available
|
|
|
41
|
13
|
M
|
W
|
L
|
|
Pedestrian/
|
No X-rays available
|
+
|
|
|
|
|
|
|
|
MVA
|
|
|
|
23
|
14
|
M
|
W
|
R
|
8
|
MVA
|
L frontal SAH/contusion diffuse SAH
|
|
|
|

ID Time after TBI

1 73 days
2 24 months
3 51 days
4 27 days
5 101 days
6 19 months
7 136 months
8 89 days
9 148 days
10 46 days
11 67 days
12 213 days
13 59.5 months
14 42 days
15 39 months
16 7 months
17 48 days
18 149 months
19 27 days
20 49 days
21 67 days
22 48 months
23 300 months
24 37 days

B = black; CC = confused and combative; DC = decerebrate; GCS = Glasgow coma scale; GSW = gunshot wound;
H = hispanic mixed race; L = left; MVA = motor vehicle accident; R = right; SAH = subarachnoid haemorrhage; SDH = subdural haemorrhage; UC = unconscious; W = white. *GCS score at 3 days.
target on the computer
screen. The target appeared following one of four possible interval delays after the previous response, each used randomly
25% of the time (0.5, 1.0, 1.5 and
2.0 s). The target was a sharply demarcated black dot which appeared in one of 16 dot positions in random order, with location counterbalanced. The dot remained on the
screen until the subject responded. Performance was measured
as the mean reaction time across 64 trials. One secondary
task required the subject
to count aloud from one to 10 repeatedly, at a self-selected rate. The other secondary task
was
an oral digit span task, using the subject’s
own digit span to
calibrate difficulty across subjects. This was determined to be the largest
digit span which the subject was able to perform correctly
three times consecutively, without failing
three times consecutively.
Stroop Test
Subjects were required to perform two tasks: naming the colours of rectangles and naming the ink colours
in which
different colour names were printed (Stroop, 1935). For the latter task the colour names and the ink colours
were always in conflict. Performance was defined as the time required to complete each form of the Stroop Test.
Spatial delayed-response task
A
delayed-response spatial location task similar to that
developed by Funahashi et al. (1989) and used by Luciana
et al. (1992) was used. Subjects
were required to recall the
location of a black dot on a computer
monitor after a brief delay. While the subject was observing
a central fixation point, a visual stimulus
appeared for 0.2 s at a peripheral
location on an imaginary circle on the screen. This stimulus was presented within 10° of the fixation
point (to avoid the subject’s
blind spot), excluding locations of 0, 90, 180 and
270° (to avoid referencing to the exact vertical
and horizontal). After the presentation of the stimulus
the screen was blank
for 8 s, after which there was an auditory tone which prompted the subject to identify the location of the stimulus by touching a point on the screen. This location
was marked by movement of the cursor. Performance was defined as the
mean distance in pixels between
the stimulus and the response over the 40 trials.
Wisconsin Card Sorting Test
Subjects performed a modified
version of the Wisconsin
Card Sorting Test (Nelson, 1976).
Subjects were asked to sort cards
into different preset categories, which changed throughout the test, using only a ‘right’ or ‘wrong’ feedback after each card placement. The sequence of categories was always
the same: colour, then shape, then number.
The category was changed after six correct consecutive trials. The cards used were only those 24 (repeated continuously) that shared no more than one
attribute with any given card, following Nelson’s variation, so that each card placement could be interpreted unambiguously.
Subjects continued until they had either achieved six categories or placed 48 cards. Performance was measured as the number
of perseverations (cards
placed in the same wrong category as an immediately preceding incorrect response).
Verbal span task
Verbal span was assessed
using a reading
span test (Daneman and Carpenter, 1980), in which subjects read through a series of sentences
which increased from two to five
sentences, remembering the last word in each sentence.
Trailmaking test
Subjects were timed while drawing a line joining
consecutive circles randomly arranged
on a page. Two different sequences were used, sequential numbers
only (Trails A) and sequential numbers and letters,
which the subject had to connect in an
alternating pattern (Trails B). Errors were pointed out as
they
occurred, according to Reitan’s method (Lezak, 1995),
so that the patient could always complete
the test. This method penalizes for errors indirectly through increased time
requirement, and the scoring is based on time alone.
Controlled oral word association test (FAS Test) Subjects named all the words
they could that began with a
given letter of the alphabet in three 1-min trials (Lezak,
1995). The testing used two sets of letters, one for each testing session, which
were balanced for frequency (F, A and
S for
the first set, and C, L and T for the second set).
Control tasks
Subjects performed a bi-letter cancellation test, similar
to that used by Luciana et al.
(1992), in which they read through six rows of letters and crossed out all of the Es and Cs.
Performance was measured as the time required to complete
this task and the number of errors made. In addition,
several of our tasks had baseline
conditions (simple visual reaction time alone for the dual-task
paradigm, colour-naming
control task for the Stroop Test and trailmaking
A time for the trailmaking
test) which should also assess basic arousal
and speed of perceptual
registration, cognitive processing and motor
response.
For the purpose of evaluating
the effect of dopamine
manipulation on cognitive function,
we grouped the tasks
according to different component
processes that may be subserved by the prefrontal
cortex. Thus, our tasks were grouped into (i) those that tapped the ability to maintain
information in working memory buffers
(verbal span task and
spatial delayed-response task) and (ii) those that tapped executive control processes (dual-task paradigm, Stroop Test,
trailmaking test, Wisconsin Card Sorting Test and the FAS
Test). Finally, a third group
of tasks were those not considered
to be dependent on prefrontal
cortex function (single-task conditions from the dual-task paradigm, the Stroop control
condition, the trailmaking A condition and the bi-letter cancellation test).
Data analysis
Overall comparisons of performance on and off bromocriptine were done with three repeated measures
MANOVA for the groups of cognitive tasks described above, with drug condition and specific individual test measures as within-subject factors and drug order as a between-subject factor. These statistical tests were performed with ranked data since raw data violated the assumptions of univariate and multivariate
homogeneity of variance (Akritas, 1991). Data were ranked separately
for each task across both drug conditions. Bonferroni correction of three
MANOVAs leads
to a conservative alpha level
of
0.0167. We used Koch’s
(1972) adaptation of the Mann–
Whitney U statistic (which
allows for potential
period effects and non-parametric data) to perform
post hoc testing of the

 Table 2 Effects of bromocriptine on cognitive task performance in TBI patients
on drug versus placebo, and comparison
with a group of control subjects not taking medication
Table 2 Effects of bromocriptine on cognitive task performance in TBI patients
on drug versus placebo, and comparison
with a group of control subjects not taking medication

 Task Measurement n Placebo Drug Mann– P-value Control*
(mean) (mean) Whitney (mean)
Task Measurement n Placebo Drug Mann– P-value Control*
(mean) (mean) Whitney (mean)
Central executive system
Dual task: counting Reaction time decrement (ms) 22 198 96 27 0.028 39
Dual task: digit span Reaction time decrement (ms) 21 539 400 21 0.016 200
Trailmaking test Reaction time decrement (s) 23 83 64 22.5 0.013 23.5
Stroop interference test Reaction time decrement (s) 23 38 33 33.5 0.05 26
FAS Test Words produced 23 31 35 27.5 0.02 54
Wisconsin Card Sorting Perseverations (number) 20 2.9 1.7 23 0.041 2.1
Working memory
|
Spatial delayed response task
|
Distance error (pixels)
|
22
|
30
|
29
|
|
Reading span
|
Total correct words
|
19
|
38
|
37
|
|
Control
|
|
Dual task: baseline
|
Reaction time (ms)
|
22
|
391
|
429
|
|
Stroop colour control
|
Task time (s)
|
23
|
67
|
65
|
|
Trailmaking A
|
Task time (s)
|
24
|
45
|
48
|
|
Letter cancellation test
|
Task time (s)
|
19
|
166
|
181
|
|
|
‡ ‡ 20
‡ ‡ †
‡ ‡ 303
‡ ‡ 45
‡ ‡ 25
‡ ‡ †

P-value represents drug versus placebo comparison. *Control
data from previous
study (McDowell et al.,
1997). †Tests not used in previous study. ‡Univariate post hoc statistical tests were not warranted in cognitive domains where the MANOVA was not statistically
significant.
effect of bromocriptine on specific tests, because the data
were not normally distributed.
Koch’s procedure consists of ranking the period differences for all of the patients
in the trial and then using the Mann–Whitney U test for the
differences between the two sequence groups. The use of a test which specifically examines period effects
ensures that significant differences are not attributable to anything that may
cause a general
change between test sessions, such as
practice effects or drug carryover
effects.
For those tasks which had more than one condition
(dual- task, Stroop and trailmaking), performance was characterized
with respect to two broad dimensions:
baseline task performance and decrement
on the second condition
compared with baseline. Performance
on each of these tasks was
measured by response
time; the performance decrement was therefore calculated as the difference between speeds on the secondary and the corresponding baseline tasks (Table 2), and
these decrement scores were used in the MANOVAs and post hoc testing. Simple differences
were an appropriate calculation since the performance decrement had been found to be unrelated to
baseline
speed
in
control
subjects
in
a previous study (McDowell et al., 1997). Speeds were characterized by mean performance times, which are sensitive
to variable and outlier data that reflect inconsistent performance after TBI.
Results
Overall behavioural performance compared with
controls
A previous study
which compared cognitive performance
on
most of the same measures between TBI patients and demographically matched control subjects
showed significant impairment in
cognitive
performance
after
TBI
using a
multivariate statistical analysis (McDowell et al., 1997).
Post hoc comparisons of individual tests showed that individuals
with TBI had impaired cognitive
function as measured
by all of the tests but the Wisconsin
Card Sorting Test, after Bonferroni correction for multiple statistical testing. Since that study included
21 of
the 24 TBI patients in this study, and the mean performance
data are similar or slightly worse
in this study, the performance
impairment in TBI patients remains (Table 2).
Tests of executive
control
The
effect of bromocriptine on performance for these
cognitive tests was examined
with a repeated measures MANOVA (Fig.
1). There was a large drug condition effect [F(1,13) = 10.033, P = 0.007], but no significant order effect [F(1,13) = 0.107, P = 0.749] and
no
significant
Drug X Order interactions [F(1,13) = 4.673, P = 0.050]. However,
the Drug X Test
interaction
was significant [F(5, 65) = 3.921, P = 0.014], showing that performance
on certain measures was more significantly
improved by bromocriptine than on others.
In fact, the effect size of the differences ranged from 0.34 for the Stroop Test to 0.71 for
the trailmaking test. Post hoc univariate analyses confirmed a beneficial effect of bromocriptine on the dual-task,
the trailmaking test, the Stroop
Test, the Wisconsin
Card-Sorting Test and the FAS Test (Table 2). The average
performance improvement in
these executive control
tests was just >25%.
Tests of working memory maintenance
The
effect of bromocriptine on the performance of tasks
which seem to require only the maintenance of information without any active
manipulation of stored
information was
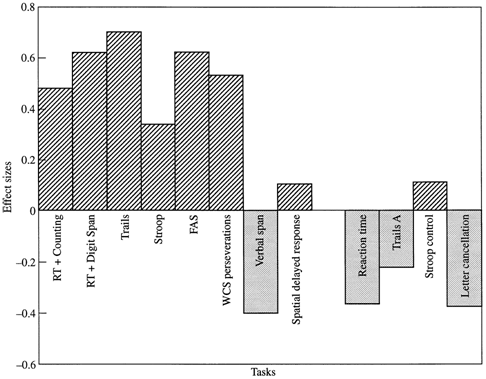
Fig. 1 Effect of bromocriptine on different tasks. For each task, this chart shows the effect size of the performance change with
bromocriptine [mean of (drug – placebo)/standard deviation
of (drug – placebo)], a positive value indicating improved performance on the drug. Effect size was used only for graphical
purposes to allow a comparison
of many different tasks on the same scale. Since most
measures are time-related, where greater values indicate
worse performance, the difference was calculated as the placebo
measurement minus the drug measurement, although
this difference was reversed (drug measurement minus placebo measurement) for the FAS Test
and the spatial delayed-response task, for which greater values indicate better performance, to maintain consistency of direction with the
other measures on this chart. For this graph, upward bars indicate
drug benefit. For tasks with more than one level the corresponding decrement scores were subtracted. RT = reaction time; WCS = Wisconsin Card Sorting Test.
assessed similarly to the tests above (Fig. 1). Bromocriptine had no significant effect
on these tasks [F(1,15)
= 0.001,
P = 0.978], with no significant effects of order or Drug X Order interaction. Individual
test scores are shown in Table
2,
with a non-significant trend towards
worse performance. Post hoc testing was not performed
to reduce type I error,
since the multivariate test was not significant.
Control tests
The
impact of bromocriptine on the control task and baseline
conditions of the other executive measures
was assessed. Basic attentional and sensorimotor
processes were not improved on bromocriptine (Fig. 1). Statistical tests analogous
to those performed above showed no significant main effect of the drug on these tests
[F(1,14) = 3.203, P = 0.095], although there was a trend for performance to be worse on
the medication. Neither
was there any significant order effect or DrugXOrder
interaction. Individual test scores are shown
in the table. Again, post hoc testing was not performed.
Speed–accuracy assessment
Performance on some tasks
(the bi-letter cancellation test, the Stroop Test and the dual task with concurrent digit span)
can
be measured by both time and accuracy, and both could be
affected by the medication.
To
make certain that the
beneficial effect of bromocriptine on speed for these tasks was
not due to speed–accuracy
trade-off, the effect of medication on task accuracy was also assessed
with the Wilcoxon signed rank test. Accuracy was not significantly
affected by bromocriptine for any of these tasks (Z = –1.23 to –1.85, P = 0.065–0.22), and the non-significant changes in function that did occur with bromocriptine were also in the
direction of improvement.
Discussion
The
pattern that emerges from the data is that bromocriptine, a dopaminergic agonist, generally
improved performance in individuals with TBI on tasks thought
to engage executive processes (i.e. the dual task, the trailmaking
test, the Stroop Test, verbal
fluency and the Wisconsin
Card Sorting Test).
However, performance did not improve on the baseline
condition of several of our executive measures
(i.e. dual task, trailmaking test and Stroop Test), working
memory measures with minimal
executive demands (i.e. verbal span and delayed response task), or the control task. This differential effect of bromocriptine suggests that its action
is not mediated through
a
non-specific increase in arousal,
attention or response
speed, but probably through improvement in specific
cognitive processes.
A lack of improvement on our control
measures may be somewhat surprising, because dopaminergic agents
have been used to improve
general arousal and attention
in individuals with brain damage (Van Woerkom et al.,
1982; Lal et al.,
1988). Reports of this effect, however, are mostly case reports,
not controlled studies;
also they used higher medication doses and did not strictly measure arousal to distinguish
it from other cognitive processes. In other words, the reported improvement in arousal and attention may actually be related to an improvement in prefrontal function, which is less easily defined and not often measured. Dopamine
agonists have also been used to improve motor speed in patients
with Parkinson’s disease
(Godwin-Austen and Smith,
1977), but they did not have that effect here, as demonstrated
by a
lack of improvement in speed-related
measures not dependent on executive function
(i.e. the bi-letter
cancellation task and control tasks of the Stroop, trailmaking and dual-task paradigms). This
disparity may be explained
by a differential dose effect, because Parkinson’s disease patients are usually treated with substantially
higher doses of dopamine agonists than those used in the present study, and their motor slowing may be of a different type than that seen in TBI.
Since this differential
behavioural effect on prefrontal function was unexpected,
alternative explanations should be considered. For example, these results could be due to differences in the interval after drug administration that each
of the tasks were given. However, this is unlikely
for several reasons. First, the half-life of bromocriptine, which is ~3–7 h, is longer than the time it took to complete our complete battery of tasks. Secondly, tasks
that showed a drug effect were intermixed in time with those that did not. Finally,
our statistical analysis accounted for the possibility of any significant differences between
the drug and placebo being due to general changes
between test sessions,
such as practice effects or drug carryover effects. Another possible explanation for our results is that the tasks that did not show improvement on bromocriptine were less sensitive
in the detection of
differences. Again, this is unlikely, since the range of performance on these tasks by patients was quite broad. For
example, on the spatial delayed response task, difference scores between sessions
ranged from an improvement
in spatial error of 22.7 pixels
to a decrement of 12.8 pixels
(raw data ranged from 8.8 to 68.4 pixels). Likewise, the verbal span test difference scores
ranged from an improvement of six correct words to a decrement
of 10 words (raw data ranged from 16 to 52 words recalled). Related
to this issue, it is possible
that our patients
were more impaired on those tasks that responded to bromocriptine than on tasks that
did not respond to bromocriptine. Such a task-difficulty
explanation seems unlikely
since our subjects were impaired,
relative to control subjects, on all of the tasks we administered except for the Wisconsin Card Sorting Test, and even this task showed improvement with bromocriptine.
Although it has been predicted from non-human primate studies that tasks that require the ability to maintain information in working memory, such as delayed-response
paradigms, should be
modulated by dopamine (Brozoski et al., 1979; Sawaguchi and Goldman-Rakic,
1991), the evidence from humans is less clear. Two studies from the
same laboratory have shown that performance on a spatial delayed-response task in normal
subjects, similar to that used in
non-human primates and identical
to the task used in our
study, is improved on bromocriptine (Luciana
et al., 1992; Luciana and Collins, 1997). However, in our own previous study with a group of normal subjects
(Kimberg et al.,
1997), and in the current study with frontally
damaged patients, we did not find improvements on this spatial
delayed-response task or a verbal span measure. Similarly,
a preliminary study from another group did not find
improvement on a delayed- response paradigm with bromocriptine, but they did find improvement with pergolide, an agonist of
D1 and D2
receptors (Mu¨ ller et al., 1998).
Although the literature
is slightly conflicting regarding the role
of dopamine in maintenance
processes, we believe that bromocriptine
has a
greater effect on tasks that require
executive control for successful performance. This suggests that executive control processes and maintenance
processes may be supported
by distinct neuroanatomical or neurochemical
substrates. Petrides and colleagues have proposed a model of functional organization of the prefrontal cortex that would be consistent with this hypothesis (Petrides,
1994). According to their
‘two-stage model’, there
are two processing systems, one dorsal and the other ventral, within the lateral prefrontal
cortex. It is proposed that the ventral region is the site where information is initially
received from posterior association areas and where active comparisons of information held in working memory are made. In contrast, the dorsal region is recruited only when monitoring and manipulation within working memory is required.
Consistent with this model is the demonstration that dorsal prefrontal regions are activated during spatial memory tasks that have maximal monitoring requirements
compared with a spatial memory task with minimal monitoring requirements, which activated only a ventral
frontal region (Owen et al., 1996). Furthermore, a meta-analysis of all functional neuroimaging studies of working memory
reported to date also supports this type of prefrontal organization (D’Esposito et al., 1998a)
as does a direct empirical
functional MRI test of this
hypothesis (D’Esposito et al., 1998b).
Although
there are no other empirical data suggesting that a neurochemical dissociation exists along the lines
mentioned above, there is evidence that Parkinson’s disease patients, who are dopamine-depleted, are impaired
differentially on different components of working memory. For example, Parkinson’s disease patients in the early stages of disease are impaired on spatial and not object working
memory tasks, suggesting that these different processes have different sensitivities
to dopamine depletion (Owen
et al., 1997; Postle et al.,
1997). Consistent with these findings
is the
demonstration in this study that, in normal subjects,
bromocriptine administration improved performance on a
spatial but not an object working
memory task. Moreover, haloperidol, a
dopaminergic antagonist, impaired
performance on only the spatial task (Luciana and Collins, 1997).
More pertinent to our findings, Parkinson’s disease patients
that have been studied off and on their medications
show differential impairments on prefrontal tasks comparable with
those used in our study. For example, Parkinson’s
disease patients on their dopaminergic medication (compared
with off it) have been shown
to perform better on a wide range of
executive function tasks, such as the Wisconsin
Card Sorting Task, a verbal fluency task and the Tower of London task (Bowen et al., 1975; Cooper
et al., 1992; Lange
et al., 1992, 1995).
In one study, whilst on their medication
Parkinson’s disease patients
were better on executive
measures, whereas no difference was found on a verbal and spatial short-term span test (Lange et al., 1995).
The neural
basis of the improvement on prefrontal tasks in
our patients is not entirely
clear. Although most of the primate studies link prefrontal function to D1 receptor activity, bromocriptine is a D2 agonist. That is, monkeys improve on
spatial working memory tasks when administered D1 agonists (Arnsten et al.,
1994) and are impaired
when given D1 antagonists (Sawaguchi and Goldman-Rakic,
1991; Arnsten et al., 1994). However, a recent study from the same
laboratory is conflicting and difficult to interpret—the prefrontal
cortex neurons in monkeys demonstrated increased activity during spatial working
memory tasks with iontophoretic infusion of a D1 antagonist (Williams and Goldman-Rakic, 1995). Very low doses of bromocriptine
have some D1 antagonist activity (Goetz
and
Diederich,
1992), and this D1 antagonism could be responsible for the
improvement seen on spatial delayed-response paradigms
in
humans (Luciana and Collins, 1997). This effect may be
separate from the D2 agonist effects of bromocriptine. The presence of D2 receptors in the human cortex is not well established, because very low levels of dopamine
in the cortex preclude accurate
biochemical analysis. Some studies, though, have shown stratification of D2 receptors to layer
V of the prefrontal cortex (Goldman-Rakic et
al., 1990). However,
most D2 receptors are in the striatum (Camps et al., 1989). Finally, a recent study from Goldman-Rakic’s laboratory shows that a D2 agonist can improve spatial
delayed-response task performance
in monkeys (Arnsten et al., 1995). Further
testing showed this effect to be mediated
by interactions between D1 and
D2 receptors. Therefore,
the effects of bromocriptine on our subjects could be the result of direct
prefrontal D2 receptor activity, or of the activity
of striatal D2 receptors,
which in turn activate the prefrontal
cortex through mesocortical dopaminergic
loops. Nevertheless, the relationship
between dopamine and prefrontal function,
as demonstrated in human pharmacological
studies and Parkinson’s
disease studies, seems strong.
In summary, our empirical findings have shown that dopamine appears
to modulate executive processes which
are
impaired after damage to the prefrontal cortex. This finding is consistent with a similar
study performed in our
laboratory studying the effects of bromocriptine
in normal young adults (Kimberg et al.,
1997). In that study it was
shown that the effect of bromocriptine depended
upon the working memory capacity of the individual. That is, bromocriptine was shown to improve performance on prefrontal measures in subjects who had lower working
memory capacity, whereas it impaired performance in subjects with higher working memory capacity. Our TBI patients all had low working memory capacity, and as a group they consistently improved on bromocriptine. Taken together, these
two studies highlight the spectrum of dopamine function that exists between normal
control subjects and patients with brain damage, and its link with cognitive processes
dependent on the prefrontal cortex.
Although
individuals with frontal lobe pathology
can perform well on standard
neuropsychological tests, they often exhibit significant
functional difficulties (Eslinger and Damasio, 1985; Shallice and Burgess, 1991). These practical
difficulties, which seem to be caused by deficits in
organization, planning and goal integration,
are
probably
due to impairments in the processes
that improved on bromocriptine in our study. Since incomplete recovery of prefrontal function
prevents full reintegration into society, the development
of more effective treatment approaches for these patients, such as pharmacological intervention, will be
an extremely important component of their care.
Thus, our findings provide
a foundation for potential therapies that may not only improve executive
function in patients with
prefrontal damage, but also decrease
the disability associated with these cognitive impairments.
Acknowledgements
We
wish to thank Dan Kimberg for helpful discussions of this work. This project
was
supported in part by NIH
grants HD07425, HD01097, NS01762 and AG13483, the
McDonnell-Pew Program in Cognitive Neuroscience, and by funds from the Moss Rehabilitation Research Institute.
References
Adams JH, Scott G, Parker LS, Graham DI, Doyle D. The contusion index: a quantitative approach to cerebral contusions in head injury.
Neuropathol Appl Neurobiol
1980; 6: 319–24.
Adams JH, Graham DI, Murray LS, Scott G. Diffuse axonal injury due to non-missile head injury in humans: an analysis
of 45 cases. Ann Neurol 1982; 12: 557–63.
Akritas MG. Limitations of the rank transform procedure:
a study of repeated
measures designs, Part I. J Am Stat Assoc 1991;
86:
457–460.
Arnsten KT, Cai JX, Murphy BL, Goldman-Rakic PS. Dopamine
D1 receptor mechanisms in the cognitive
performance of young
adult and aged monkeys.
Psychopharmacology (Berl) 1994; 116:
143–51.
Arnsten AF, Cai JX, Steere JC, Goldman-Rakic PS. Dopamine D2 receptor mechanisms contribute to age-related cognitive decline:
the effects of quinpirole on memory and motor performance in monkeys. J Neurosci 1995; 15: 3429–39.
Baddeley A. Working
memory. [Review]. Science 1992; 255: 556–9. Baddeley A,
Della
Sala
S,
Papagno
C,
Spinnler
H.
Dual-task
performance in dysexecutive and nondysexecutive patients
with a
frontal lesion. Neuropsychology 1997; 11: 187–94.
Bench CJ, Frith CD, Grasby PM, Friston KJ, Paulesu E, Frackowiak
RS, et al. Investigations of the functional anatomy of attention using the Stroop Test. Neuropsychologia 1993; 31: 907–22.
Berman KF, Ostrem
JL, Randolph C, Gold J, Goldberg TE, Coppola R, et al. Physiological activation of a cortical network during performance of the Wisconsin Card Sorting Test: a positron emission tomography study. Neuropsychologia 1995; 33: 1027–46.
Bowen FP, Kamienny RS, Burns
MM,
Yahr M. Parkinsonism:
effects of levodopa treatment on concept formation. Neurology
1975; 25: 701–4.
Brown RM, Crane AM, Goldman PS. Regional distribution of monoamines in the cerebral cortex and subcortical structures of the rhesus
monkey: concentrations and in vivo synthesis rates.
Brain Res 1979; 168: 133–50.
Brozoski TJ, Brown RM, Rosvold
HE, Goldman PS. Cognitive deficit caused by regional
depletion of dopamine
in prefrontal cortex of
rhesus monkey. Science 1979; 205: 929–32.
Camps M, Corte´s R, Gueye B, Probst A, Palacios
JM. Dopamine receptors in human brain: autoradiographic distribution of D2 sites. Neuroscience 1989; 28: 275–90.
Cicerone KD. Attention deficits and dual task demands
after mild traumatic brain injury. Brain Inj 1996; 10: 79–89.
Cooper JA, Sagar HJ, Doherty SM, Jordan N, Tidswell P, Sullivan EV. Different effects of dopaminergic and anticholinergic therapies on cognitive and motor function
in Parkinson’s
disease. Brain 1992;
115: 1701–25.
D’Esposito M, Detre JA, Alsop DC, Shin RK, Atlas S, Grossman
M. The neural basis of the central
executive system of working memory. Nature 1995; 378: 279–81.
D’Esposito M, Aguirre GK, Zarahn EK, Ballard D, Shin RK, Lease
J. Functional MRI studies of spatial and non-spatial
working memory. Cog Brain Res. In press 1998a.
D’Esposito M, Postle BR, Ballard D, Lease J. Maintenance
versus manipulation of information held in working memory: an event-
related fMRI study. Brain and Cognition.
In press 1998b.
Daneman M, Carpenter PA. Individual
differences in working memory and reading. J Verb Learn Verb Behav 1980; 19: 450–66.
de
Keyser J, Ebinger
G, Vauquelin G. Age-related changes in the human
nigrostriatal dopaminergic system. Ann Neurol 1990; 27:
157–61.
Divac L, Rosvold
LE,
Szwarcbart
MK.
Behavioral
effects
of
selective ablation of the caudate nucleus. J Comp Physiol Psychol
1967; 63: 184–90.
Durso R, Fedio P, Brouwers
P, Cox C, Martin AJ, Ruggieri
SA, et al. Lysine vasopressin in Alzheimer’s disease.
Neurology 1982;
32: 674–7.
Eslinger PJ, Damasio AR. Severe disturbance of higher cognition after bilateral frontal lobe ablation: patient EVR. Neurology
1985;
35: 1731–41.
Funahashi S, Bruce
CJ,
Goldman-Rakic PS. Mnemonic coding of visual
space
in
the
monkey’s dorsolateral prefrontal
cortex.
J Neurophysiol 1989; 61: 331–349.
Funahashi S, Bruce CJ, Goldman-Rakic PS. Dorsolateral
prefrontal lesions and oculomotor delayed-response performance: evidence
for mnemonic ‘scotomas’. J Neurosci 1993; 13: 1479–97.
Fuster JM, Alexander GE. Neuron activity related to short-term memory. Science 1971; 173: 652–4.
Godwin-Austen RB, Smith NJ. Comparison of the effects of
bromocriptine and levodopa in Parkinson’s
disease. J Neurol Neurosurg Psychiatry 1977; 40: 479–82.
Goetz CG, Diederich NJ. Dopaminergic agonists for the treatment of Parkinson’s disease.
[Review]. Neurol Clin 1992; 10: 527–40.
Goldman-Rakic PS, Lidow MS, Gallager
DW. Overlap of dopaminergic, adrenergic, and serotoninergic receptors and
complementarity of their
subtypes
in
primate
prefrontal
cortex.
J Neurosci 1990; 10: 2125–38.
Hartman A, Pickering RM, Wilson BA. Is there a central executive
deficit after severe head injury? Clin Rehabil 1992; 6: 133–40.
Jonides J, Smith EE, Koeppe RA, Awh E, Minoshima
S, Mintun MA. Spatial working memory in
humans as revealed by PET [see
comments]. Nature 1993; 363: 623–5. Comment in: Nature
1993;
363: 583–4.
Kimberg DY, Farah MJ. A
unified account of cognitive impairments following frontal damage: the role of working
memory in complex organized behaviour. J Exp Psych: Learn Mem Behav 1993; 122:
411–28.
Kimberg DY, D’Esposito M, Farah MJ. Effects of bromocriptine
on human subjects depend on working memory capacity. Neuroreport
1997; 8: 3581–5.
Koch GG. The use of non-parametric methods in the statistical analysis of the two-period change-over design. Biometrics 1972;
28: 577–84.
Lal
S, Merbtiz CP, Grip JC. Modification of function in head- injured patients with sinemet
[see comments]. Brain Injury 1988;
2: 225–33. Comment in: Brain 1989; 3: 319–22.
Lange KW, Robbins TW, Marsden CD, James M, Owen AM, Paul GM.
L-dopa withdrawal in Parkinson’s
disease selectively impairs cognitive performance in tests sensitive
to frontal lobe dysfunction.
Psychopharmacology (Berl) 1992; 107: 394–404.
Lange KW, Paul GM, Naumann M, Gsell W. Dopaminergic effects of cognitive performance
in
patients
with
Parkinson’s disease. J Neural Transm Suppl 1995; 46: 423–32.
Lezak MD. Neuropsychological
assessment. 3rd ed. New York:
Oxford University Press; 1995.
Luciana M, Collins PF. Dopaminergic
modulation of working memory for spatial but not object cues in normal humans. J
Cogn Neurosci 1997; 9: 330–47.
Luciana M, Depue RA, Arbisi P, Leon A. Facilitation of working memory in humans by a D2 dopamine
receptor agonist. J Cogn
Neurosci 1992; 4: 58–68.
McDowell S, Whyte J, D’Esposito M. Working memory impairments in traumatic brain injury: evidence from a dual-task
paradigm. Neuropsychologia 1997; 35: 1341–53.
Milner B. Effects of different brain
lesions on card sorting. Arch
Neurol 1963; 9: 90–100.
Mu¨ ller U, von Cramon DY, Pollmann
S. D1 versus D2-receptor modulation of visuo-spatial working memory
in humans. J Neurosci
1998; 18: 2720–8.
Nelson HE. A modified
card sorting test sensitive to frontal lobe defects. Cortex 1976; 12: 313–24.
Owen AM, Evans AC, Petrides M. Evidence
for a
two-stage model of spatial working memory processing within the lateral frontal cortex: a positron emission tomography study. Cereb Cortex 1996;
6:
31–8.
Owen AM, Iddon JL, Hodges JR, Summers
BA, Robbins TW. Spatial and non-spatial
working memory at different stages of
Parkinson’s disease.
Neuropsychologia 1997; 35: 519–32.
Petrides M. Frontal
lobes
and
behaviour. [Review]. Curr Opin
Neurobiol 1994; 4: 207–11.
Postle BR, Jonides J, Smith EE, Corkin S, Growdon JH. Spatial,
but not object, delayed response
is impaired in early Parkinson’s disease. Neuropsychology 1997; 11: 171–9.
Rinne JO, Lonnberg P, Marjamaki P. Age-dependent decline in human
brain dopamine D1 and D2 receptors. Brain Res 1990; 508:
349–52.
Sawaguchi T, Goldman-Rakic PS. D1 dopamine receptors
in prefrontal cortex: involvement in working memory. Science 1991;
251: 947–50.
Shallice T. Specific impairments of planning. Philos
Trans R Soc
Lond B Biol Sci 1982; 298: 199–209.
Shallice T, Burgess PW. Deficits
in strategy application following frontal lobe damage in man. Brain 1991; 114: 727–41.
Stroop JR. Studies of interference in serial verbal reactions. J Exp
Psychol 1935; 18: 643–62.
Taylor AE, Saint-Cyr JA, Lang AE, Kenny FT. Parkinson’s
disease and depression. A critical re-evaluation. Brain 1986; 109: 279–92.
Van
Woerkom TC, Minderhoud JM, Gottschal
T, Nicolai G.
Neurotransmitters in the treatment
of patients with severe head injuries. Eur Neurol 1982; 21: 227–34.
Vendrell P, Junque C, Pujol J, Jurado MA, Molet J, Grafman J.
The role of prefrontal regions
in the Stroop task.
Neuropsychologia
1995; 33: 341–52.
Verin M, Partiot A, Pillon B, Malapani
C, Agid Y, Dubois B.
Delayed response tasks and prefrontal lesions in man—evidence for self generated
patterns of behaviour with poor environmental modulation. Neuropsychologia 1993; 31: 1379–96.
Williams GV, Goldman-Rakic PS. Modulation of memory fields
by dopamine D1 receptors
in prefrontal cortex [see comments]. Nature
1995; 376: 572–5. Comment in: Nature 1995; 376: 549–50.
Wong
DF, Wagner HN Jr, Dannals
RF,
Links JM, Frost JJ, Ravert HT, et al. Effects of age on dopamine and serotonin receptors measured by positron
tomography in the living human
brain. Science
1984; 226: 1393–6.
Zarahn E, Aguirre GK, D’Esposito M. Delay-specific activity within prefrontal cortex demonstrated during a working
memory task: a functional MRI study. Soc Neurosci Abstr 1996; 22: 968.
Received January 5, 1998. Accepted January 21, 1998
![]()
![]()
